Migraines, Acupuncture and Weapons of Mass Destruction

The Glamorous History of Botox for Migraines
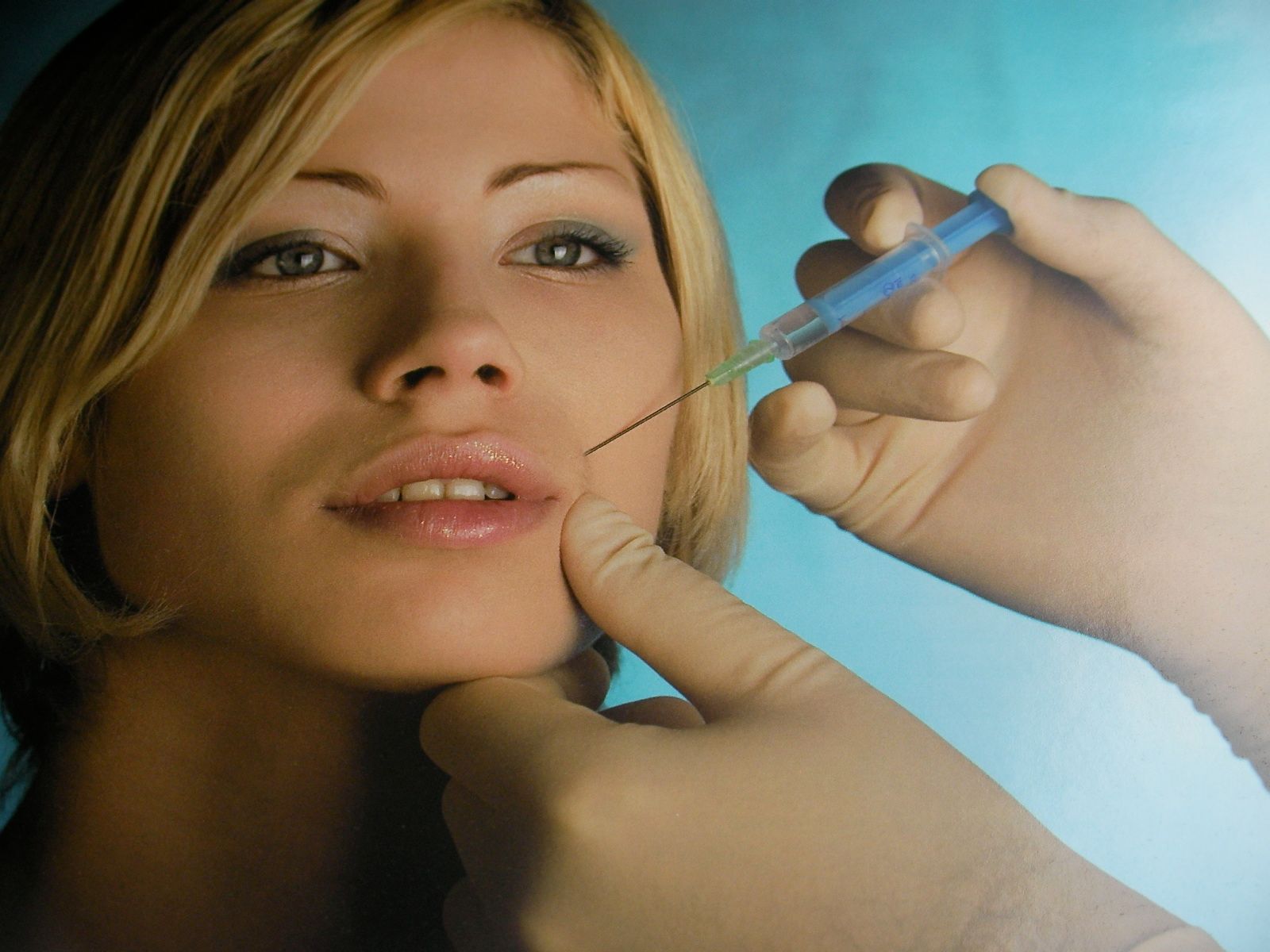
Like many important discoveries, this one happened quite by accident . . .
In the early 1990s, most people agreed that electively having a toxic nerve agent injected into your face was probably a pretty bad idea on the whole, even if it did smooth out your wrinkles. But by the eve of the new millennium, attitudes had done an about face and people were lining up left, right and centre to have the treatment done. (And last year, it was the most popular cosmetic procedure performed in the United States, outnumbering breast augmentation, nose reshaping, eyelid surgery, liposuction and facelift combined. 1)
Yes, I’m talking about the injection of botulinum toxin type A, aka Botox, a “purified” version of the most acutely lethal poison known to man.
Headaches and Wrinkles Be Gone
As hordes of people stampeded to iron out their wrinkles, the cosmetic surgeons performing the technique started to notice an interesting trend in the feedback from their patients: people who had formerly suffered with chronic headaches and migraines were now becoming free of this ailment. The injection of Botox into the face, it would seem, was taking away the headaches along with the crow’s-feet.
Unsurprisingly, this was very welcome news for Allergan, the manufacturer of Botox. For naturally with more clinical indications comes more sales. And so, Allergan set out to fund a series of studies, to provide evidence for a new clinical use for Botox.
First, an open-labeled study in 1998 confirmed that headaches were indeed reduced after treatment with Botox.2 But how would it stand up compared to placebo control?
Well, to be honest the results were mixed and mostly negative. After nearly a decade of funding studies, it looked like Botox didn’t outperform saline injections for the treatment of tension-type headache nor did it help most migraine sufferers and it had a lot more side-effects. 3 It’s not that people didn’t get better after the treatment, mind you. Large improvements were seen whether the injection contained the active treatment or not.
Not to be discouraged, Allergan dug deep and identified what it hoped would be a group of people that it could show would benefit from the noxious treatment. Preliminary evidence suggested a possible treatment effect in people who suffered more than 15 headaches per month and it was on that sub-population that Allergan would focus its efforts.
Preemptive Measures
The Phase III REsearch Evaluating Migraine Prophylaxis Therapy (PREEMPT) trials were the largest to date. Identical in design, the multi-centred twin studies ran from 2006 to 2008. When the results of PREEMPT 1 were published in 2010, the disappointment must have been unbearable. “No significant between-group difference for onabotulinumtoxinA versus placebo was observed for the primary endpoint, headache episodes (-5.2 vs. -5.3; p 1⁄4 0.344). Large within-group decreases from baseline were observed for all efficacy variables.” 4 In other words, people got significantly better whether or not they were injected with the saline or the poison. Secondary outcome measures did show some statistically significant, albeit small, effects, but that wasn’t gonna cut it.
While the study protocol for PREEMPT 2 had been settled years earlier and the data already collected by the time the disappointing results from PREEMPT 1 were published, desperate times called for desperate measures. The researchers at Allergan scrambled to change the main outcome measure from headache episodes to frequency of headache days before unblinding the data, and this time they achieved a small, but statistically significant positive effect.5
A Bit of Misdirection?

As exciting as it must have been to finally have large-scale randomized trials to support the use of Botox for migraines, a few niggles remained unresolved. For one, blinding wasn’t very good in the studies because side effects were so much more common in the Botox group, that people knew if they were getting the real deal or not, and this could explain the difference between the two groups.
As headache experts Jes Oleson and Peer Tfelt-Hansen remarked: “no assessment of the effectiveness of blinding was done. Blinding seems likely to have been far from optimal . . . This presumed lack of blinding alone can probably explain a 10% difference between the active drug and placebo.” 6
There was also the nagging “mechanism” issue – supposedly, Botox prevented migraines by blocking pain signals through nerves by blocking the release of acetylcholine. But this didn’t make sense considered in light of the fact that it didn’t work for tension type headache or episodic migraines (migraines occurring fewer than 15 days a month).
And finally, the really consistent and impressive improvement in the group receiving saline injections despite the poor blinding was a real head scratcher indeed. “Although the combined results for headache days were significant, the difference between the active and placebo groups was only around 10% for most variables and there was no difference between groups in the reduction of intake of acute medicines. The published papers give much emphasis to the difference between baseline and treatment, which is indeed impressive, but is equally so in the placebo group. . . .”7
These weaknesses not withstanding, Allergan requested and received licensing approval in the UK and the US to market Botox for the prophylaxis of chronic migraines in 2010. Allergan then submitted a request to NICE for a Health Technology Assessment, which was published in the summer of 2012. It presented its own systematic review, which consistented entirely of the two PREEMPT trials. It submitted its own safety review, and by sticking solely to data gathered from RCTs it was able to avoid mention of adverse effects and the dozens of deaths noted in the post-licensure literature (who knew that Botox could cause botulism?) 89 and to down play the ironic fact that a significant side effect of Botox for migraine prevention is actually the triggering of migraines.
A few months later, NICE published its new guidelines on the management of headaches and included a qualified recommendation for Botox. While the Scottish Medicines Consortium rejected the use of Botox for chronic migraines in 2011 and again in 2013 due to a perceived poor cost benefit ratio, overall Allergan had done a decent job of getting the regulatory authorities behind its product under the cirucmstances.
And this is all fine, as long as no one mentioned the long established treatment of migraines with the use of needles alone.
It’s the needles, stupid!

For not once in the pharmaceutically funded discussion of unknown mechanisms or larger than expected placebo effects is the copious research literature on acupuncture for the treatment of migraines considered. But just as I like to remind my cat when her head is under the blanket, “just because you can’t see me doesn’t mean that I can’t see you,” just because you haven’t acknowledged the evidence-base for using needles sans neurotoxin as an effective treatment for migraines, doesn’t mean that it’s not there.
In fact, it’s not often that a large pharmaceutical company provides such high quality, large-scale evidence for the efficacy of acupuncture! Thank you, Allergan!
Acupuncture & Botox – A Head to Head Comparison
So after mulling over the dubious background of Botox for migraines, how does acupuncture compare in terms of evidence base, effectiveness compared to drugs, cost-effectiveness and recommendations?
Well, back in 2004, eight years before Allergan made its pitch to NICE, an NHS Health Technology Assessment undertaken for NICE found that compared to controls, patients who had acupuncture used 15% less medication, made 25% fewer visits to GPs and took 15% fewer days off sick. 10 Meanwhile, the PREEMPT studies shows no difference in medication use between Botox and placebo.
In 2009, a Cochrane Systematic review, the highest quality evidence summary available, concluded that “Available studies suggest that acupuncture is at least as effective as, or possibly more effective than, prophylactic drug treatment, and has fewer adverse effects.” 11 The only systematic reviews performed on Botox for prophylaxis of chronic migraines were performed by Allergan itself and fly a bit fast and loose with their interpretations of the data. But overall, studies find that Botox is equal to prophylactic drugs in both effectiveness and side-effects 12. Acupuncture has been found to be superior on both accounts.
And while the most recent NICE guidelines recommend Botox when a) patients have tried three other medications b) have headaches more than 15 days a month and c) are not overusing painkillers, these same guidelines suggest a course of 10 acupuncture treatments for anyone who has migraines (episodic or chronic) where topiramate and propranolol are unsuitable or ineffective.
So, in a nutshell, the NICE guidelines for the management of migraines make a stronger recommendation for the use of acupuncture that applies to more people than the Botox recommendation. The respective NHS health technology assessments find that acupuncture is more cost-effective than Botox.13 The difference in safety profile is beyond refute. Acupuncture is safer, more cost-effective, and has much stronger evidence for its use.
Is the NHS addicted to drugs?

Side by side, there’s really no comparison between acupuncture and Botox. In fact, I hate to point out the painfully obvious, but no proponent of Botox has ever acknowledged the fact that as an intervention, it’s more likely than not just poisonous acupuncture. That not withstanding, by every relevant measure, acupuncture should be a promoted and recommended treatment for migraine sufferers to at least the same extent as Botox, and arguably more so.
Let’s be NICE
Unfortunately, just because NICE takes the time and resources to publish clear, evidence-based guidelines doesn’t mean that they’re actually put into practice. Implementation depends on a number of factors, including the opinions of key influential players, as we will see.
The Migraine Trust
According to its website, The Migraine Trust is “the health and medical research charity for migraine in the United Kingdom . . . [It] seeks to empower, inform, and support those affected by migraine while educating health professionals and actively funding and disseminating research.”14 Groovy, so what empowering words of wisdom do they have about treatment options for migraineurs?
Well, on the Treatment page, in addition to the standard drugs, they mention Botox for migraines and a link to a Factsheet dedicated to the subject. They also mention Biofeedback, a treatment that didn’t make it into the NICE guidelines. And acupuncture? Not mentioned once.
The website also contained some other interesting nuggets, including a strong level of support from Dr Fayyaz Ahmed , a trustee of the Migraine Trust, for the use of Botox. Further searching reveals that Dr Ahmed is the foremost expert on the use of Botox for migraines in the UK, training practitioners and performing studies. He also seems to have a financial relationship with Allergan, the makers of Botox.15 – which I’m sure has nothing to do with anything and I only mention for the sake of completeness.
25 Sept 2014 – I’m backing this claim up with a declared conflict of interest statement from the above cited 2014 study: “Fayyaz Ahmed has received honorarium to deliver training workshops for Allergan paid to British Association for the study of headache (BASH) and received honorarium to attend Allergan Advisory Board meetings.” (Dr Ahmed is the Chair of BASH)
And what are Dr Ahmed’s views on acupuncture for migraine? In a 2012 review published in the British Journal of Pain, he writes: “Non-drug interventions such as herbal medication (butterbur), acupuncture, homeopathy, Indian head massage and cognitive behaviour therapy may well help although the evidence remains inconclusive.” 16
Ex-squeeze me? I don’t know about butterbur or Indian head massage, but acupuncture had the support of an NHS Health Technology Assessment and a Cochrane Systematic Review at the time he wrote that article, which is more than can be said for Botox, which he did put forth as a valid treatment option in the same article. Migraine Trust? More like migraine dis-trust. (See what I did there?)
If all that nonsense weren’t enough, Dr Ahmed takes things a step further by actually using acupuncture to sell people on Botox. In a 2012 interview for the Daily Mail, Dr Ahmed is quoted as saying about Botox, “since it’s no more painful than acupuncture, patients don’t need an anaesthetic.” 17
Whoa, whoa, hang on there, Tiger. First you slander acupuncture by making false statements about lack of evidence without so much as a reference to back it up, and now what are you on about? The administration of Botox uses a 30 gauge hypodermic needle, the diameter of which is approximately .3112mm. I can’t speak for everyone, but the needles I use in my practice are between .18 -.20 mm in diameter, which is 36 – 42% smaller. And yes, bigger needles hurt more.18 So don’t kid yourself, Dr Ahmed, in addition to being less effective and more dangerous, Botox is significantly more painful.
The British Association for the Study of Headache (BASH)
My main concern about the BASH guidelines is that the most recent version is from 2010, so they are not based on the current NICE recommendations, which were published in 2012. As such, rather than interventions being listed in order of evidence or indication, they’re listed as “pharmacological” followed by “Non-drug Intervention.” And within this subset, the order of interventions seems arbitrary as while acupuncture has the highest-level of evidence out of this group, it’s listed third.
And, I should add, it’s these 2010 guidelines that are listed on the Royal College of GPs website, describing them as: “Guidelines for all healthcare professionals in the diagnosis and management of migraine, tension-type, cluster and medication-overuse headache.” 19 Seriously? Guidelines that precede the now two year-old NICE guidelines and make no mention of them? I think we can do better.
Migraine Action
On the Migraine Action website, if we go to Information -> Treatments & Therapies -> Preventative Treatments, again we see drugs and Botox as options, but no acupuncture. There is a section on “Complementary Therapies,” which includes a leaflet about acupuncture. 20 The trouble is, if it’s a treatment recommended by the NICE guidelines, it’s no longer “complementary” to standard treatment, it is standard treatment. I think Mr Acupuncture Leaflet deserves a new home with the information about the other “regular” treatments on your website, don’t you?
Meanwhile, back at the doctor’s office

So, it seems that the major patient advocacy groups who should be informing people about their treatment options are totally dropping the ball on acupuncture while simultaneously going bananas for Botox. But what’s actually happening at the doctor’s office?
Well, it’s really difficult to get national statistics for the UK about treatments, since everyone is pretty much doing their own thing. So I can only comment on what appears to be happening in my neck of the woods.
NHS Provisioning: “Acupuncture? We don’t do that”
About a month ago, a patient came to see me for migraines. He had had them for about 30 years and he was now unemployed and on disability because he was no longer fit to work. He got on very well during our first session, and I recommended that he go back to his GP and see if he could get a referral for acupuncture. I even told him to mention to the doctor that it’s recommended in the NICE guidelines. He emailed me back a couple of days later saying that he had asked his GP about getting acupuncture on the NHS for his migraines. The doctor’s response? “We don’t do that.” Huh.
I was a bit curious if this was an isolated dearth of availability to evidence based medicine on the NHS or if this was more wide spread. A good friend of mine is a GP in a large practice in Leicestershire. The next time I saw her, I asked: “Listen, if you had a patient who was coming to see you for migraines, and both propranolol and topiramate weren’t helping, could you refer them for acupuncture?”
Her response: “Hmmm, no, I don’t think we do that.”
Me: “What about Botox?”
GP: “Well, we wouldn’t refer for a particular treatment. We would refer to the neurologist and then they would decide the best course of action.”
Me: “But say they’d already been to the neurologist and had had their scans, had ruled out anything more serious, and now they’ve come back to see you, what then?”
GP: “Well, we don’t refer for acupuncture.”
Hmmmm.
And it goes even farther up the food chain. Last year, I emailed a consultant neurologist and head of the Migraine Clinic at the Leicester General Hospital Neurology department about available treatments for migraine. His response?
“In conversation with patients if they ask me if they can try acupuncture I usually say to try it if they wish but I myself don’t formally refer patients for this.” 21 Well gosh, I didn’t realise that providing access to NICE recommended treatment was optional! Any particular reason why your patients have to pay for this standard treatment themselves if they wish to have it?
Yes, there is the odd GP who offers acupuncture in their own practice. And there may be some availability in certain parts of the country, but the accessibility is anything but uniform.
According to the Migraine Trust website: “Access to treatments that have been recommended by NICE for use in the NHS, if your doctor says they are clinically appropriate for you, is a patient right enshrined in the NHS Constitution.” Of course, the Migraine Trust are referring to Botox here, but I think we can agree that the same principle applies.
Parting Thoughts
When Botox and acupuncture go head to head in terms of evidence base, cost effectiveness and safety, there’s really no comparison. Sure, acupuncture research isn’t without it’s peccadilloes, although interestingly the issues that befall Botox research are remarkably similar. So, in terms of the merits of recommending these interventions, the indications are clear cut. Acupuncture is recommended for a larger population, is more cost effective, and has fewer side effects. Moreover, it is part of the NICE guidelines for the management of migraine.
So why we see a systematic promotion of Botox and yet a simultaneous omission of acupuncture by patient advocacy groups and the NHS I really cannot say. It may be that no one who has ever recommended Botox was able to mention acupuncture in the same sentence, lest admitting the obvious, that it’s the needles, not the Botox, that are actually causing the vast majority of the therapeutic effect. Or it may be that the head of some so called “patient advocacy groups”, the same ones who are responsible for educating GPs on the management of migraines, are putting ties with Allergan ahead of patient interests.
But while these factors may contribute, I suspect that the situation arises mostly from something much more inane and kind of boring: old paradigms die hard and the mainstream medical establishment continues to make the artificial separation between “conventional medicine” and “complementary medicine” rather than “evidence-based medicine” and other treatments, as is its mandate. Somehow sticking needles into people that are filled with a noxious substance is just more compatible with established views than using needles alone, even if the evidence clearly says otherwise.
The only way you can promote the use of Botox for migraines in terms of efficacy, cost effectiveness, and safety is if you pretend that acupuncture doesn’t exist. Period. If we can all agree that it does, and not only does it exist but we can acknowledge that it’s a treatment endorsed by the NHS’s own guidelines, then some things have really gotta change.
1 Hotta, T. A. (2014). ASAPS (American Society of Aesthetic Plastic Surgery)–2013 Annual Statistics. Plastic Surgical Nursing : Official Journal of the American Society of Plastic and Reconstructive Surgical Nurses, 34(2), 47–48. doi:10.1097/PSN.0000000000000045)
2 Binder, W. J., Blitzer, A., Schoenrock, L. D., & Pagoda, J. M. (2000). Botulinum toxin type A (BOTOX) for treatment of migraine headaches: an open-label study. Otolaryngology – Head and Neck Surgery, 123(6), 669–676.
3 Jackson, M., & Barbuto, J. P. (2008). Botox, migraine, and the American Academy of Neurology: an antidote to anecdote. Journal of Managed Care Pharmacy : JMCP, 14(5), 465–467.
4 Aurora, S. K., Dodick, D. W., Turkel, C. C., DeGryse, R. E., Silberstein, S. D., Lipton, R. B., et al. (2010). OnabotulinumtoxinA for treatment of chronic migraine: Results from the double-blind, randomized, placebo- controlled phase of the PREEMPT 1 trial. Cephalalgia : an International Journal of Headache, 1–11. doi:10.1177/0333102410364676
5 Diener, H., Dodick, D., Aurora, S., Turkel, C., DeGryse, R., Lipton, R., et al. (2010). OnabotulinumtoxinA for treatment of chronic migraine: Results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 2 trial. Cephalalgia : an International Journal of Headache, 30(7), 804–814. doi:10.1177/0333102410364677
6 Olesen, J., & Tfelt-Hansen, P. (2010). Licence for Botox in so-called chronic migraine. The Lancet, 376(9755), 1825–1826. doi:10.1016/S0140-6736(10)62165-4
7 Ibid
8 Coté, T. R., Mohan, A. K., Polder, J. A., Walton, M. K., & Braun, M. M. (2005). Botulinum toxin type A injections: Adverse events reported to the US Food and Drug Administration in therapeutic and cosmetic cases. Journal of the American Academy of Dermatology, 53(3), 407–415. doi:10.1016/j.jaad.2005.06.011
9 Commissioner, O. O. T. (n.d.). 2008 – FDA Notifies Public of Adverse Reactions Linked to Botox Use. Fda.Gov.
10 Vickers, A. J. (2004). Acupuncture for chronic headache in primary care: large, pragmatic, randomised trial. BMJ (Clinical Research Ed.), 328(7442), 744–0. doi:10.1136/bmj.38029.421863.EB.
11 Linde, K., Allais, G., Brinkhaus, B., Manheimer, E., Vickers, A., & White, A. R. (2009). Acupuncture for migraine prophylaxis (Review). Cochrane Database of Systematic Reviews, (1).
12 Ramachandran, R., & Yaksh, T. L. (2014). Therapeutic use of botulinum toxin in migraine: mechanisms of action. British Journal of Pharmacology, 171(18), 4177–4192. doi:10.1111/bph.12763
13 Royle, P., Cummins, E., Walker, C., Chong, S., Kandala, N.-B., Waugh, N., & Coventry, C. V. (2012). Evidence review group report: Botulinum toxin type A for the prophylaxis of headaches in adults with chronic migraine. NHS Reports.
14 migrainetrust.org. (n.d.). migrainetrust.org. Retrieved September 20, 2014, from migrainetrust.org.
15 Khalil, M., Zafar, H. W., & Quarshie, V. (2014). Prospective analysis of the use of OnabotulinumtoxinA (BOTOX) in the treatment of chronic migraine; real-life data in 254 patients from Hull, UK. The Journal of Headache and Pain
16 Ahmed, F. (2012). Headache disorders: differentiating and managing the common subtypes. British Journal of Pain, 6(3), 124–132. doi:10.1177/2049463712459691
17 Botox: Now on the NHS to stop migraines | Daily Mail Online. (n.d.). Botox: Now on the NHS to stop migraines | Daily Mail Online. Dailymail.Co.Uk. Retrieved September 20, 2014, from dailymail.co.uk/health/article-2171140/Botox-Now-NHS-stop-migraines.html.
18 Gill, H. S., & Prausnitz, M. R. (2007). Does needle size matter? Journal of Diabetes Science and Technology, 1(5), 725–729.
19 Headache. (n.d.). Headache. Rcgp.org.Uk. Retrieved September 20, 2014, from rcgp.org.uk/clinical-and-research/clinical-resources/headache.aspx
20 (n.d.). migraine.org.uk. Retrieved September 21, 2014, from migraine.org.uk/information/treatments-and-therapies/preventative-treatments/#preventative
21 Anonymous. (2013, October 21). Email from Dr Anon, private communication.

